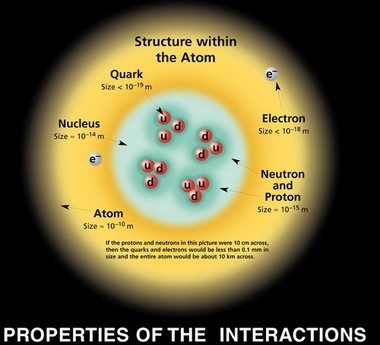
When looking at the standard atom model (left), it is easy to assume that protons, neutrons, and electrons are all relatively the same size. Though easy to assume based on looking at this model of an atom, it is completely wrong. Really, protons and neutrons are significantly larger than electrons. "Significantly" means like 10,000 times smaller (at largest) than protons and neutrons. Protons and Neutrons are already 100,000 times smaller than the actual atom, and electrons are 100,000,000 times smaller than the atom, so the atom is mostly empty space (as in 99.999999999999% empty space).
