This is an introduction to how elements form compounds and why.
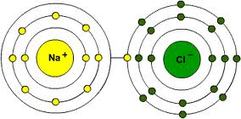 This is an illustration of Sodium Chloride.
This is an illustration of Sodium Chloride.
Chemists' work with an atoms electrons a whole lot: the electrons control the reactivity of the atom. Different groups on the periodic table are known for being either more reactive or less reactive or willing to form compounds. Alkali Metals, in particular, are known to be extremely volatile. The reason? Alkali metals have exactly one extra electron in it's outer shell, and they want to get rid of that one electron at all costs so they can be a happy, stable eight valence electron elements. Another notorious group of elements are the halogens, which have the opposite problem: halogens have seven electrons in their outer shell and desperately want one more electron to be happy, stable eight valence electron elements. When left together they form a happy, stable compound (NaCl or Sodium Chloride). Now that they have formed their compound, however, they cannot separate they are locked into formation. Sodium Chloride is also known as table salt. An atom's goal is to have a full outer electron shell. (The Noble Gases, however, already have a full outer shell so they don't need or want to form compounds.)
This is a picture of the sodium lending chlorine the electron it needs.You could say this relationship "binds them together."
This is a picture of the sodium lending chlorine the electron it needs.You could say this relationship "binds them together."
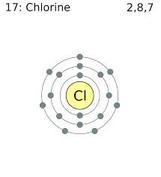
Every element forms several different compounds (excluding the noble gases, and a few of the other more standoffish elements) Chlorine one of the more notorious of the bunch, you may just not know it. You may have heard that Chlorine was used as a poison gas in the chemical warfare of World War I. You may have not known that this has everything to do with molecules and forming compounds. When you inhale deadly chlorine gas it literally rips the hydrogen out of the water (H20) and mucus in your lungs to get the extra electron it wants. This form HCl (Hydrochloric acid) and HOCl (Hypochlorous acid). Hypochlorous acid is relatively mild, but hydrochloric acid is extremely corrosive. Chlorine gas causes intense burning and irritation as well as breathing difficulty (because you are not inhaling oxygen). This particular chemical reaction and compound formation causes a rather painful way to die.
