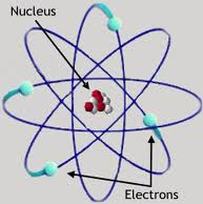
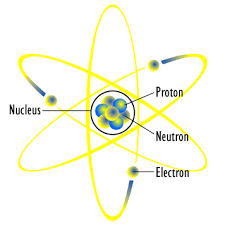
Changing an Atom

Protons. Protons determine the atomic number of an element which holds the entire identity of the element. Simply if the number of protons in a nucleus changes, then the entire element changes. If you have Beryllium (4) and you take away a proton the atomic number is now 3 so the element is Lithium (3).
Beryllium turns into lithium (but not a very stable lithium, because you're just changing the protons... the picture on the right is not completely accurate in this case.)

Neutrons. To find the number of neutrons in an atom, you must subtract the atomic mass (the mass of the neutrons and protons in the nucleus of a regular atom of that type) from the atomic number (the number of protons in a nucleus of an atom). The number of neutrons is not the same as the protons! In Carbon there are six neutrons regularly in the nucleus, called Carbon-12 (the number of protons and neutrons combined). If you add two more neutrons to Carbon-12, it becomes unstable Carbon-14 used in carbon dating. Think of isotopes as different versions of an element. If you add or subtract enough neutrons from an element, all of them can become radioactive (elements that are radioactive naturally just means that they can't become stable). Just neutrons are neutral it doesn't mean that they don't make a difference.

Electrons. When you loose or gain electrons the atom stops being neutral and becomes an ion. An Ion has an electric charge, ions can either be +ion or -ion. A positive ion is called a cation. Anions are negative ions. Having a net negative charge means you have too many electrons, having a net positive charge means you are missing electrons. Example: Lithium(3) has a +2 charge how many electrons does it have? The answer is 1, because lithium is element number 3 so it needs 3 electrons to be neutral. If it is missing two electrons (because protons charge is equal to electron charge so it needs two negative to make it neutral [two electrons]) and 3 (the number of electrons there should be) - 2 (the number it needs) = 1 (the number it has).