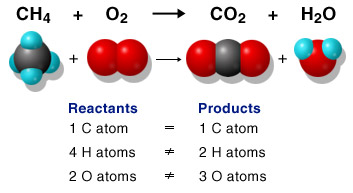
Balancing a chemical equation
THIS PAGE IS UNDER CONSTRUCTION. I apologize for the inconvenience.
If you haven't read and understood "The Great and Powerful Chemical Equation," it would be beneficial for you to look over it to ensure that you understand the components and vocabulary on this subject.
A chemical equation always must be balanced. Because of the Law of the Conservation of Mass ("Matter can be neither created or destroyed") an unbalanced equation must be altered.
If you haven't read and understood "The Great and Powerful Chemical Equation," it would be beneficial for you to look over it to ensure that you understand the components and vocabulary on this subject.
A chemical equation always must be balanced. Because of the Law of the Conservation of Mass ("Matter can be neither created or destroyed") an unbalanced equation must be altered.
Simple Example Equation:
02Ge2+H = 0Ge+H
02Ge2+H = 0Ge+H
|
Changing an equation can be done only by changing coefficients to make the reactant and product have the same number of atoms, just rearranged. Below is a step-by-step guide to solving a chemical equation.
STEP 1: Make a list of the number of each type of atom on each side of the yield sign. This is pictured to the right. In this case the list would look like this: The reactants would be listed as: Germanium: 2 Oxygen: 2 Hydrogen: 1 The products would be listed as: Oxygen: 1 Germanium: 1 Hydrogen: 1 The purpose of these lists is to help you keep track of how many atoms are on each side. You will be changing these numbers as soon as you add coefficients to the values in an attempt to balance it. When the equations are balanced, the two lists should have identical values for each element. The first letter in the atomic symbol of an element is always capitalized, so any lower case letter is part of the chemical symbol starting with the uppercase letter. O2Ge2 is a compound containing two oxygen atoms and two germanium atoms (this is just an example). |