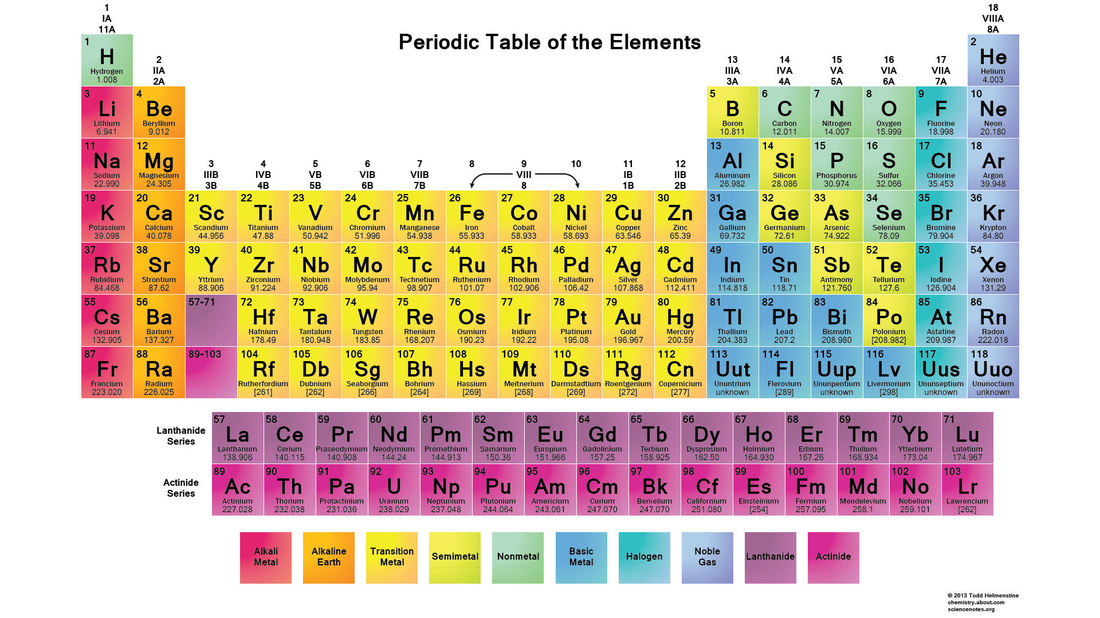
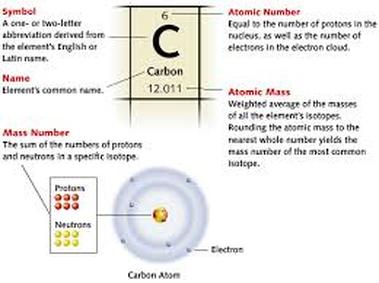
This is the display chart for everything that you have ever heard of (that is tangible)... what does it mean?
1. Colors: Each of the colors on this chart are representing a group of elements (the key is at the bottom). Each group of elements has to do with their properties; for example: on the far right column of the table (the Noble Gases) all of these elements are gases and are completely inert and do not willingly form compounds (though in extreme conditions, like hundreds of degrees below zero, we have convinced all of the Noble Gases except for Neon and Helium to form extremely unstable and wobbly compounds), because of their completely full shell of electrons. These similarities have helped scientists categorize these elements into what we call the periodic table of the elements. The colors vary based on
2. The Boxes: So you have probably figured out that each of these boxes represents each element, but there are a lot of different things in each block. Using the example of Silicon(14) here is how it works:
2a: The big letters in the middle of the block are the atomic symbols of the elements, usually the letters in the name of the element (similarly to the United States's state abbreviation, like Alaska's abbreviation is AK). Silicon's atomic symbol is Si. The catch: not all symbols reflect the name of the element, some of the symbols reflect the Latin name for the element, like Tantalum's "W'. These atomic symbols are used in compound formulas such as NaCl, or Sodium Chloride, or table salt.
2b. The numbers in the top left of the element block is the atomic number, or the number of protons in the nucleus of the atom. Silicon's atomic number is 14.The number of neutrons can change (to create different isotopes of an element, see "And Everything Smaller" for more information on Neutrons), or the number of electrons can change (to create negative or positive ions, see "And Everything Smaller" for more info on electrons) but if the number of protons changes then the element changes. The number in parentheses after an element is listed in text is usually the atomic number. See "Changed: Ions and Isotopes" for more information on what happens when you take away certain particles.
2c. The name on the second-to-last line on the element block is the name of the element. Believe it or not, Silicon's name is really Silicon... shocker, right? The name is what we call the element, but in all the name is really not any where near the most important piece of information on the element block. At the bottom of the main table, in the far right, there are several elements with three letter abbreviations and with names like Ununoctium. These names are just stand in names while the naming committee argues over what the name should be. Many of the transuracnic (the elements after Uranium, 92.) are named after chemists or physicists (and a few after other non-chemist-or-physicist notables in science, Nobelium and Copernicium, for example), though not the ones that discovered them... odd how that works.
2d. The number at the bottom of the element tile is usually the atomic mass in amu. Atomic mass is the average weight of the atom, measured in atomic mass units (amu). Rounding this number to the nearest whole number will give you the number of protons and neutrons in an atom. To calculate the number of neutrons you must subtract the atomic number from the atomic mass (more information on the "Changed: Ions and Isotopes"). This number is a decimal because the weight of protons is only 99.86% the mass of a neutron (Electrons are 0.054% a neutrons mass). Silicon's atomic mass is 28.086 amu.
3. The numbers on two sides of the periodic table are there to be used a coordinates to indicate certain elements. Referring to blocks in a horizontal row you use the word periods (replacement for "row" when referring to the periodic table), going vertically the columns are groups (as opposed to columns). (Osmium's is group 8, period 6).
1. Colors: Each of the colors on this chart are representing a group of elements (the key is at the bottom). Each group of elements has to do with their properties; for example: on the far right column of the table (the Noble Gases) all of these elements are gases and are completely inert and do not willingly form compounds (though in extreme conditions, like hundreds of degrees below zero, we have convinced all of the Noble Gases except for Neon and Helium to form extremely unstable and wobbly compounds), because of their completely full shell of electrons. These similarities have helped scientists categorize these elements into what we call the periodic table of the elements. The colors vary based on
2. The Boxes: So you have probably figured out that each of these boxes represents each element, but there are a lot of different things in each block. Using the example of Silicon(14) here is how it works:
2a: The big letters in the middle of the block are the atomic symbols of the elements, usually the letters in the name of the element (similarly to the United States's state abbreviation, like Alaska's abbreviation is AK). Silicon's atomic symbol is Si. The catch: not all symbols reflect the name of the element, some of the symbols reflect the Latin name for the element, like Tantalum's "W'. These atomic symbols are used in compound formulas such as NaCl, or Sodium Chloride, or table salt.
2b. The numbers in the top left of the element block is the atomic number, or the number of protons in the nucleus of the atom. Silicon's atomic number is 14.The number of neutrons can change (to create different isotopes of an element, see "And Everything Smaller" for more information on Neutrons), or the number of electrons can change (to create negative or positive ions, see "And Everything Smaller" for more info on electrons) but if the number of protons changes then the element changes. The number in parentheses after an element is listed in text is usually the atomic number. See "Changed: Ions and Isotopes" for more information on what happens when you take away certain particles.
2c. The name on the second-to-last line on the element block is the name of the element. Believe it or not, Silicon's name is really Silicon... shocker, right? The name is what we call the element, but in all the name is really not any where near the most important piece of information on the element block. At the bottom of the main table, in the far right, there are several elements with three letter abbreviations and with names like Ununoctium. These names are just stand in names while the naming committee argues over what the name should be. Many of the transuracnic (the elements after Uranium, 92.) are named after chemists or physicists (and a few after other non-chemist-or-physicist notables in science, Nobelium and Copernicium, for example), though not the ones that discovered them... odd how that works.
2d. The number at the bottom of the element tile is usually the atomic mass in amu. Atomic mass is the average weight of the atom, measured in atomic mass units (amu). Rounding this number to the nearest whole number will give you the number of protons and neutrons in an atom. To calculate the number of neutrons you must subtract the atomic number from the atomic mass (more information on the "Changed: Ions and Isotopes"). This number is a decimal because the weight of protons is only 99.86% the mass of a neutron (Electrons are 0.054% a neutrons mass). Silicon's atomic mass is 28.086 amu.
3. The numbers on two sides of the periodic table are there to be used a coordinates to indicate certain elements. Referring to blocks in a horizontal row you use the word periods (replacement for "row" when referring to the periodic table), going vertically the columns are groups (as opposed to columns). (Osmium's is group 8, period 6).
Updating the periodic table happens every once and a while. They may add a new element or update some of the most recent element discovery's name (the Unun... names are just place holders while the committee argues over which name that new element should get). The version above is the most updated version so far.
Wow, did you really read all that? Good job. Very few pages will have this much text, most will have more diagrams.
Wow, did you really read all that? Good job. Very few pages will have this much text, most will have more diagrams.